Rare Nephrology News
Advertisement
Spotlight On
X-Linked Hypophosphatemia (XLH)
X-linked hypophosphatemia, is an X-linked dominant form of rickets that differs from most cases of rickets in that vitamin D supplementation does not cure
Prevalence
1-9 / 1,000,000
Age of Onset

ICD-10
E83.3
Inheritance
Autosomal dominant

Autosomal recessive

Mitochondrial/Multigenic

X-linked dominant

X-linked recessive

Rare View
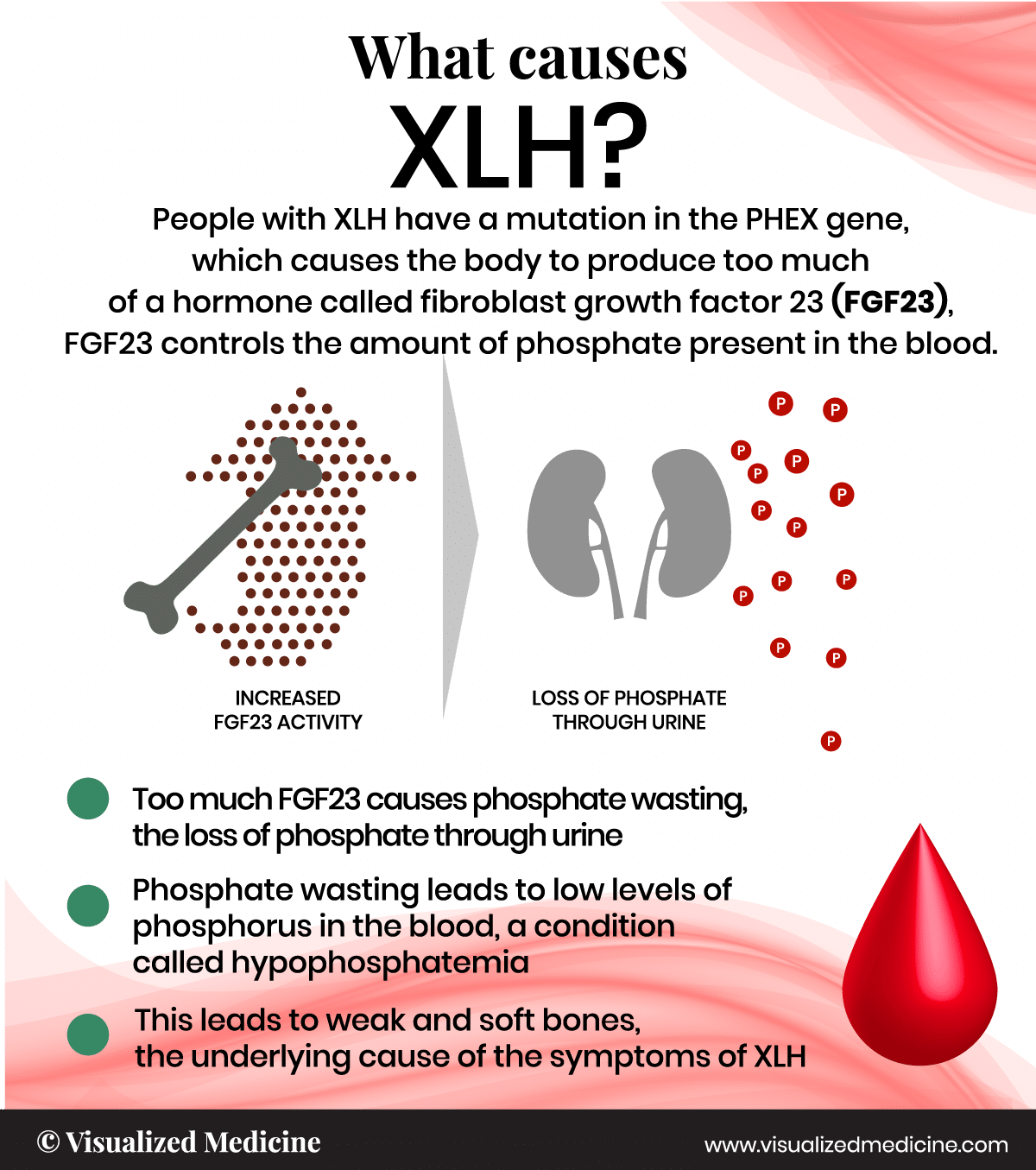
XLH, or X-linked hypophosphatemia, is a condition that affects bones, muscles, and teeth due to the excessive loss of phosphate. Phosphate is lost through the urine, which causes low levels of phosphorus in the blood, a condition called phosphate wasting or hypophosphatemia. Rickets is a hallmark of XLH and is caused by the softening of bones during growth in childhood. Rickets is a key feature of XLH in children and causes symptoms such as bowed legs.†
5 Facts you should know
FACT
An inherited disorder characterized by low levels of phosphate in the blood.
FACT
Phosphate levels are low because phosphate is abnormally processed in the kidneys, which causes a loss of phosphate in the urine (phosphate wasting) and leads rickets.
FACT
XLH is usually diagnosed in childhood.
FACT
XLH is caused by mutations in the PHEX gene on the X chromosome, and inheritance is X-linked dominant.
FACT
Treatment generally involves supplements of phosphate and high-dose calcitriol and may also include growth hormones, corrective surgery, and dental treatment.
Interest over time
Google searches
Common signs & symptoms
Abnormality of dental enamel
Abnormality of the metaphysis
Abnormality of the wide portion of a long bone
Bone pain
Genu varum
Hypophosphatemia
Low blood phosphate level
Joint dislocation
Joint dislocations
Recurrent joint dislocations
Osteomalacia
Softening of the bones
Rachitic rosary
Rickets
Weak and soft bones
Tooth abscess
Current treatments
Burosumab (Brand name: Crysvita)
Manufactured by Ultragenyx Pharmaceutical, Inc.
FDA-approved indication: April 2018 approved for the treatment of X-linked hypophosphatemia (XLH) in adult and pediatric patients 1 year of age and older.
Top Clinical Trials
| Title | Description | Phases | Status | Interventions | More Information |
|---|---|---|---|---|---|
| Open Label Trial Assessing Safety and Efficacy of Burosumab (KRN23), in a Patient With ENS and Hypophosphatemic Rickets | A 52 week, open label trial to assess the safety and efficacy of KRN23, an investigational antibody to FGF23, in a single pediatric patient with Epidermal Nevus Syndrome(ENS) and associated hypophosphatemic rickets | Completed | Biological: Burosumab | Phase 3 | More Info |
| Effectiveness of Paricalcitol in Reducing Parathyroid Hormone (PTH) Levels in X-linked Hypophosphatemic Rickets | The purpose of this study is to determine the effectiveness of paricalcitol, a form of synthetic vitamin D, in lowering parathyroid hormone (PTH) levels and reducing disease symptoms in children and adults with X-linked hypophosphatemic (XLH) rickets. | Completed | Drug: Paricalcitol|Other: Placebo | Phase 3 | More Info |
| Study of KRN23 in Adults With X-linked Hypophosphatemia (XLH) | The primary efficacy objective of this study is to establish the effect of burosumab treatment compared with placebo on increasing serum phosphorus levels in adults with XLH. | Completed | Biological: burosumab|Other: Placebo | Phase 3 | More Info |
| Open Label Study of KRN23 on Osteomalacia in Adults With X-linked Hypophosphatemia (XLH) | The primary objective of this study is to establish the effect of KRN23 treatment on improvement in XLH-associated osteomalacia as determined by osteoid volume (osteoid volume/bone volume, OV/BV). | Completed | Biological: burosumab | Phase 3 | More Info |
| Efficacy and Safety of Burosumab (KRN23) Versus Oral Phosphate and Active Vitamin D Treatment in Pediatric Patients With X Linked Hypophosphatemia (XLH) | The primary objective of this study is to evaluate the effect of KRN23 (burosumab) therapy in improving rickets in children with XLH compared with active control (oral phosphate/active vitamin D). | Completed | Biological: burosumab|Drug: Oral Phosphate Supplement|Drug: active vitamin D | Phase 3 | More Info |
| Evaluation of Safety, Tolerability, and Efficacy of INZ-701 in Adults With ENPP1 Deficiency | The purpose of this study is to assess the safety and tolerability of INZ-701, an ectonucleotide pyrophosphatase/phosphodiesterase 1 (ENPP1) enzyme replacement therapy, for the treatment of ENPP1 Deficiency (including Generalized Arterial Calcification of Infancy Type 1 [GACI] and Autosomal Recessive Hypophosphatemic Rickets Type 2 [ARHR2]). | Recruiting | Drug: INZ-701 | Phase 1|Phase 2 | More Info |
Top Treatments in Research
| Agent | Class/Mechanism of Action | Development Status | Company | Clinical Studies | More Information |
|---|---|---|---|---|---|
| Drug: Paricalcitol|Other: Placebo | Paricalcitol is a synthetic, biologically active vitamin D analog of calcitriol with modifications to the side chain (D2) and the A (19-nor) ring. Preclinical andin vitro studies have demonstrated that paricalcitol's biological actions are mediated through binding of the VDR, which results in the selective activation of vitamin D responsive pathways. Vitamin D and paricalcitol have been shown to reduce parathyroid hormone levels by inhibiting PTH synthesis and secretion. | Phase 3 | Yale University|National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS) | Click here for clinical studies | More Info |
| Drug: INZ-701 | Native ENPP1 is bound to the cell membrane with the active enzymatic portion of the protein outside the cell. We fused this domain with an antibody Fc fragment to construct INZ-701. In contrast from native ENPP1, INZ-701 is a soluble protein designed to circulate throughout the body and cleave extracellular ATP into PPi and AMP, a precursor of adenosine. | Phase 1|Phase 2 | Inozyme Pharma | Click here for clinical studies | More Info |
† Genetic and Rare Diseases Information Center (GARD) - PO Box 8126, Gaithersburg, MD 20898-8126 https://rarediseases.info.nih.gov